Now Reading: Peanut Allergies: New Skin Patchimmunotherapy Treatments Shows Promise In Treatment
-
01
Peanut Allergies: New Skin Patchimmunotherapy Treatments Shows Promise In Treatment

Peanut Allergies: New Skin Patchimmunotherapy Treatments Shows Promise In Treatment
Recent research revealed that a new skin patch can be effective in treating peanut allergies. Every year approximately 3.2 million Americans suffer from peanut allergies, and few treatment options prove to be effective. In fact, the US Food and Drug Administration (FDA) has not approved any drugs for treating this condition.
Modern therapies such as immunotherapy are being developed that can mitigate potentially life-threatening allergic reactions. These therapies work by slowly decreasing a person’s sensitivity to peanut protein. Recent research revealed that a new skin patch can be effective in treating peanut allergies. A lot of research is still required on the effectiveness of thisese treatments, but early results do look positive. BothSome therapies companies have already gotten “breakthrough therapy designation” from the FDA – meaning that these treatments represent what the FDA calls a significant “unmet medical need”.
Allergies occur when the body’s immune system identifies an allergen (in this case, the peanut protein) as a foreign invader or parasite, and starts attacking those harmless proteins – it is essentially an overreaction of the immune system. You may wonder why people are purposefully exposed to substances or proteins they are allergic to when using immunotherapy, but it is a scientifically proven method of teaching the immune system to remain inactive in the presence of allergens. This is done by exposing a person to small amount of an allergen through different means. This technique is referred as allergy shots immunotherapy– in which small amounts of the proteins is delivered in pollen, pet dander, or insect stings to decrease allergen-sensitivity in a person; .this can be done through injections, through the mouth, under the tongue and on the skin through patches.
Doctors avoid peanut allergy shots because of possible life-threatening reactions. Dietary intake of a legume by an allergic person can cause nausea, hives and vomiting in mild cases. In severe cases, life-threatening anaphylactic reactions are also possible which can lead to an obstruction of an airways and ultimately death.
Companies have taken advantage of this allergy anxiety. Recent research revealed that children who consume peanuts at a young age, are less likely to be affected with the disease, which led to some companies selling expensive peanut butter, designed to prevent allergies from developing. Mylan, another pharmaceutical company, was punished for increasing the price of life-saving EpiPens. These are epinephrine injection which are helpful in decreasing the symptoms of an allergic reaction, giving a patient more time to reach the hospital for proper treatment.
Avoiding all foods containing peanuts is currently the only way to prevent allergic reactions in a control peanut allergic individualies. But food labels are not always accurate, and an allergy-sufferer can’t take risks with unknown food. But modern treatments have provided some relief to the situation.
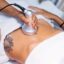
Skin patches are being developed by the French biopharmaceutical company DBV Technologies, called the “Viaskin Peanut”. This patch slowly delivers a small amount of proteins through the skin. Old patches need to be replaced after 24 hours, with new patches then applied to a new spot.
A recent year-long study, conducted by the Consortium of Food Allergy Research (CoFAR) and sponsored by the National Institutes of Health (NIH), found that almost half of the participants using this skin patch were able to increase the amount of peanut they could eat without having an allergic reaction ir peanut threshold by ten-fold. Comparatively, only about 12 percent of participants using the placebo patches could have that much amount of protein at the end of the same year. According to the findings published in the Journal of Allergy and Clinical Immunology, children between the ages of 4 and 11 years haves shown the best results.
These wearable skin patches have their own side-effects. Around 80 percent of the participants in the treatment group complained about rashes and itching under the patch. One person withdrew from the study because of complaint of severe skin irritation. “One of the things we were a little bit surprised about, but also very excited about, was that adherence was very high,” says the study’s lead author Stacie Jones, an allergist at Arkansas Children’s Hospital. “We had more than 97 percent of the participants across all the treatment groups that stayed with the study and changed the patch every 24 hours.”
Oral capsule AR101, manufactured by California-based company Aimmune is another wonderful advancement in this field. Though it is still undergoing trials, its results so far are promising. The capsule, which contains peanut proteins, has to be taken daily. It is similar to the Viaskin as it also works by providing tiny increasing doses of peanut protein. This helps to teach the body’s immune system not to react to the protein. In other words, it is decreasing sensitivity to an allergen. Significant differences between the patch treatment and oral treatment these two treatments are that an allergic person has to take AR101 orally and the dose enhances slowly through the period of 20 weeks.
In one small clinical trial conducted by Aimmune, 23 participants took AR101 daily for about 22 weeks. Six patients withdrew because of problems related to the gastrointestinal tracts. Results of the study revealed that 78 percent of the participants increased amount of peanut they could eat without having an allergic reaction increased their protein threshold by 10-fold. This means that they were able to eat 10 times more peanut protein than they could before undergoing the treatment. Participants with placebo patches could not eat that amount of protein.
Both therapies are very encouraging but further research is required says Dr Gerald Nepom, an immunologist at the Benaroya Research Institute in Washington State. The FDAAD has neither approved the patch nor the pill, but both therapies have recently completed Phase II clinical trials that were done to evaluate the productivity and safety of the treatment. Larger Phase III trials will be done for both treatments to assess their efficacy on a larger scale – with about 500 patients already screened by DBV and 500 anticipated to register for Aimmune. Results will be revealed after the second half of 2017.
It is significant to evaluate the productivity and safety of the treatment on a large scale because we should not give false hope to the affected people, says Mary Rozenman, Aimmune’s senior vice president for corporate development and strategy. “We wouldn’t want anyone to have a sense of protection who wasn’t really protected,” she says.
For the future, it is very important to amalgamate these modern therapies with immunosuppressant drugs to reduce severe reactions to peanut protein, Dr Nepom says. Lab techniques and methodologies have advanced enough to assist scientists in having a detailed picture of the body’s immune system against specific allergens. Dr Nepom was hopeful that there will be many new treatment options available in future for cure of peanut allergy.
“Now, there are really good new methodologies for interrogating the immune system and the response to peanut,” he says. “So I’m pretty optimistic actually that things are going to move a lot faster now.”